
Principal Investigator: Prof. Maja Weisser & Dr. Getrud Mollel
Project leader/ Coordinator: Dr. Ezekiel Luoga
Project Administrator: TBC
Funding Partner: European Union under the Global Health EDCTP3 Programme
Start date: July 27, 2023
End date: July 31, 2028
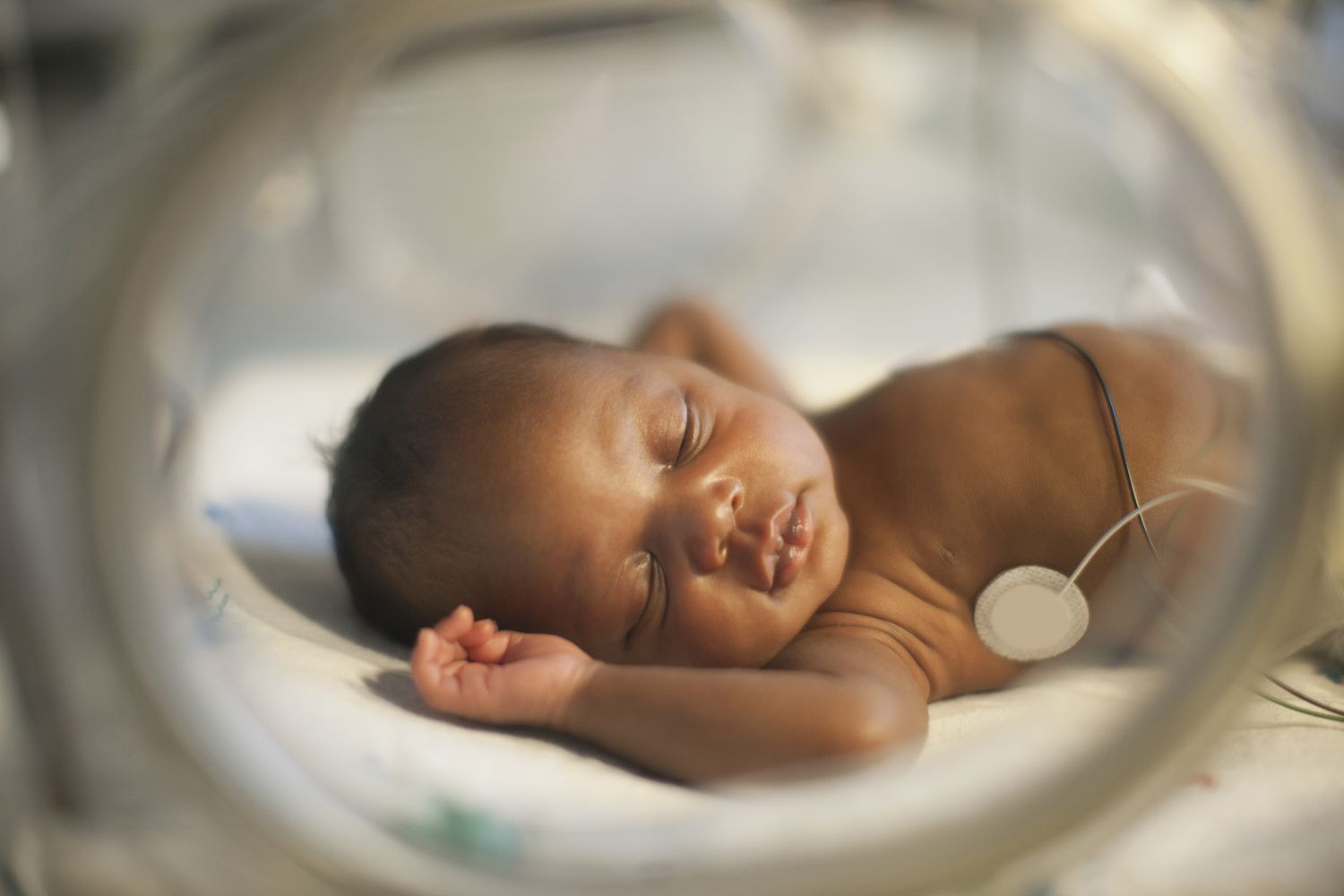
Severe neonatal infection adaptive platform trials in Africa (SNIP-AFRICA)
The Severe neonatal infection adaptive platform trials in Africa (SNIP-AFRICA) is an African-European collaboration project that aims to improve the treatment of severe infections in newborns. Using an innovative approach, the project will tackle questions around antibiotic management of neonatal sepsis in Africa, in a time of rising antimicrobial resistance.
Funded by the European Union under the Global Health EDCTP3 Programme, SNIP-AFRICA will conduct an adaptive trial to identify the best drug regimens and doses for difficult-to-treat infections and sepsis, which threaten the lives of newborns in neonatal units in sub-Saharan African countries.
Launched in July 2023 and ongoing until 2028, the five-year project brings together a consortium of 10 partners from African and European countries. The SNIP-AFRICA project is coordinated by Fondazione Penta ETS in Italy and St George’s, University of London in the United Kingdom.
Project Partners
Ifakara Health Institute is part of the SNIP-AFRICA consortium and will be collaborating in conducting an adaptive trial and interventional studies across neonatal units in Ghana, Kenya, South Africa, Tanzania and Uganda, with the objective of identifying optimal treatment regimens and doses for difficult-to-treat infections and sepsis. The trial will enroll 1,200 neonates with the first patients expected to be enrolled in June 2025.
Other partners involved in the project are University College London, UK; African Research Collaboration for Health, Kenya; Stellenbosch University, South Africa; Kwame Nkrumah University of Science and Technology, Ghana; DNDi-GARDP Southern Africa NPC, South Africa; MUJHU Care, Uganda; Ifakara Health Institute, Tanzania; and University of Antwerp, Belgium.
Ifakara will be partnering for several work packages in collaboration with the St. Francis Referral Hospital in Ifakara. Ifakara scientists who will be working on the SNIP-AFRICA project include Dr. Getrud Mollel, Prof. Maja Weisser, and Dr. Ezekiel Luoga - all site Principal Investigators.
SNIP-AFRICA Objectives
SNIP-AFRICA's primary objective is to build a sustainable platfrom to implement adaptive trials for severe childhood infections. The platform design will allow researchers to adjust the trial as it progresses based on the results of early data. In comparison to traditional trial designs, using adaptive platform trials can address multiple research questions simultaneously, providing a more personalised approach to researching neonatal sepsis.
To guarantee the sustainability of the SNIP-AFRICA platform, the Consortium will also provide training opportunities on innovative clinical trials and neonatal sepsis, building the capacity of African researchers and clinicians to develop and implement future adaptive trials.
Overall, SNIP-AFRICA’s broader goal is to improve treatment of neonatal sepsis and reduce neonatal mortality, enhance stakeholder engagement to ensure the successful implementation of adaptive trials relevant to local needs, including addressing major infection threats to children through a combination of surveillance and intervention studies.
