
Principal Investigator: Dr. Ally Olotu
Project leader/ Coordinator: Dr. Moshi Moshi Shabani
Project Administrator: TBC
Funding Partner: Novartis
Start date: April 1, 2024
End date: June 30, 2024
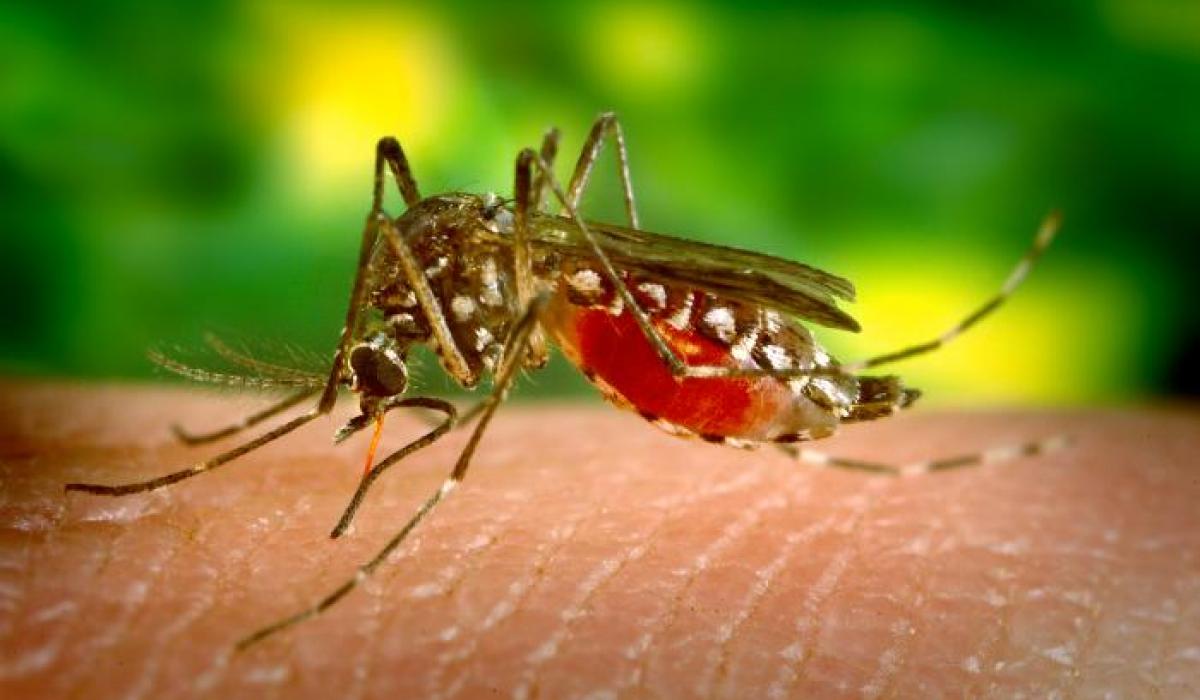
Investigating the efficacy and safety of combination drug KLU156 for treating uncomplicated malaria - “KALUMA Project"
The KALUMA project, conducted by the Ifakara Health Institute in collaboration with key partners, seeks to generate crucial insights into the efficacy, safety and tolerabilty of the combination drug called KLU156 in treating uncomplicated malaria.
The goal is to address the urgent need for effective anti-malarial treatments using combination drugs across diverse populations, aligning with broader strategies to expand health interventions in malaria-endemic regions.
Supported by Novartis, the Phase 3 study comprises a Core phase and an Extension phase, aiming to evaluate the efficacy, safety, and tolerability of KLU156 in treating uncomplicated malaria in adults and children weighing 5 kg or more.
Introduction and Rationale
All new malaria treatments should be developed as combinations of drugs with distinct mechanisms of action for synergistic or additive efficacy to mitigate the development of drug resistance and ensure cure (WHO 2022, FDA 2016a).
In the KALUMA Project, KLU156, a fixed dose combination of ganaplacide (KAF156) with lumefantrine-SDF, will be assessed.
The project will be a multicenter, open-label, randomized, non-inferiority trial conducted across various regions in Sub-Saharan Africa, including Tanzania.
The KALUMA project will involve a Core phase lasting 43 days and an Extension phase spanning 24 months. Patients will be randomly selected and enrolled into treatment to receive KLU156 during the Core phase, followed by the Extension phase with each malaria episode treated for 3 days, followed by scheduled follow-up visits.
Study Objectives
The primary objective of the Core phase is to confirm KLU156's efficacy against uncomplicated malaria, demonstrating non-inferiority to Coartem (artemether/lumefantrine). This will be assessed by Adequate Clinical and Parasitological Response (ACPR) rates at Day 29. The Extension phase will include assessing safety, tolerability, and long-term efficacy over a longer period, for up to two years.
Participants will include male and female patients aged 2 months and older, weighing at least 5 kg, with confirmed uncomplicated malaria. Approximately 1,500 patients will be randomized across 21 plus countries in Africa.
In Tanzania, the study will target enrollment of children under 12 years in areas such as in Bagamoyo, Tanga, Korogwe, leveraging a network of health facilities to facilitate recruitment, community engagement, and follow-up. Ifakara will target children below 12 years residing in Bagamoyo districts in Chalinze council.
Core and Extension phase activites
During the Core phase, patients will receive either KLU156 or Coartem. KLU156 will be administered once daily for 3 days, while Coartem will be given twice daily. The Extension phase will exclusively use KLU156. Additional treatments, including standard care and rescue medications, will be provided as needed.
Comprehensive safety assessments, including physical examination, laboratory assessments, vital signs, and ECG assessments, will be conducted throughout the study. Serious adverse events will be closely monitored and reported.
Efforts will focus on recruiting participants competitively across multiple sites with high malaria prevalence, . The study will also involve numerous health facilities and community networks, with in-house observation and follow-up visits planned across various centers.
Ifakara to lead project in TanzaniaIfakara Senior Research Scientist, Dr. Ally Olotu will lead the study as the Principal Investigator. Ifakara will target children below 12 years residing in Bagamoyo districts in Chalinze council.
Ifakara to lead project in Tanzania
Ifakara Senior Research Scientist, Dr. Ally Olotu will lead the study as the Principal Investigator, working alongside Dr. Moshi Moshi Shabani, the study Project Leader.
